
News and Announcements from the CDB
The Drosophila tracheal system is a branched network of epithelial tubes extending throughout the body to transport oxygen to the tissues. During tracheal development, the tracheal lumen is filled with apical extracellular matrix (ECM), which is later cleared out and replaced by gases prior to larval hatching. Components of the apical ECM, such as proteins and chitin, which is also an important component of the exoskeleton protecting the fly’s exterior, play important roles in tracheal morphogenesis by regulating the length and diameter of the tubes. One ECM protein required for tracheal development is a chitin deacetylase, serpentine (Serp). Past studies have reported that overexpression or loss of serp leads to overelongated and convoluted tracheal tubes. While tracheal cells are the major source of ECM proteins, including Serp, secreted into the lumen, it remains unclear whether and to what extent other tissues contribute to tracheal tube development.
Now, new work by Bo Dong and colleagues in the Laboratory for Morphogenetic Signaling (Shigeo Hayashi, Team Leader) reveals the Drosophila fat body, a mesoderm-derived tissue that plays physiological roles similar to the vertebrate liver, as a surprising new source of luminal Serp during tracheal morphogenesis. Published in the journal Development, their findings demonstrate that a non-epithelial tissue plays a role in tracheal morphogenesis and provides additional evidence of the fat body supporting the developmental process of various organs.
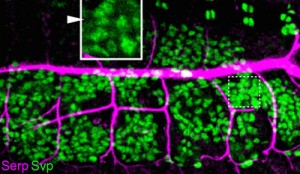
The insect fat body functions as a site of synthesis and storage of sugars, lipids and proteins, which are secreted into the hemolymph to be delivered to tissues and organs of the body as needed to maintain homeostasis. Dong et al. first confirmed the presence Serp in the fat body as well as in the tracheal system of fly embryos, consistent with previous reports. Then, using two different mutant fly strains (rab9 and shrub) in which glitches in vesicular mechanisms affect Serp localization resulting in phenotypes similar to serp mutants (i.e. overelongated tracheal tubes), they found that both mutants showed higher levels of Serp in the fat bodies and lower Serp levels in the tracheal lumen than seen in wildtype, a pattern that was not observed with other apical ECM proteins. Additional experiments specifically blocking endocytic or exocytic pathways of fat bodies indicated that the source of Serp accumulation in the fat bodies of the mutants was cell-intrinsic synthesis, ruling out the possibility that Serp was being translocated there from an external source.
Under normal conditions, Serp does not remain localized in the fat body, so the group next examined whether Serp synthesized by the fat body can be delivered to the lumen of the developing tracheal tube. They ectopically expressed a secreted luminal marker protein containing the chitin-binding domain of Serp in the fat body and found that the marker accumulated in the tracheal lumen. In contrast, when a functionally inert secreted protein marker with similar characteristics to Serp was expressed, it was absent from the lumen. Further, the authors used the technique of IR-laser induced local heat shock (Miao and Hayashi, 2014) to demonstrate that Serp-GFP could be translocated to the lumen even after the developing tracheal tube acquired epithelial barrier function, suggesting that Serp is taken up by tracheal cells from the hemolymph and deposited in the lumen via transcytosis.
Finally, the group looked at whether fat body-derived Serp plays a functional role in tracheal development. When Serp was ectopically expressed in the fat body of serp mutants, they observed that the overelongated tube phenotype could be rescued, suggesting that Serp secreted from the fat body plays a role in tracheal morphogenesis. Closer examination also revealed that the function of fat body-derived Serp appears to overlap with that of tracheal cell-derived Serp and supports tracheal tube development when the tracheal source is limited.
“To find that the fat body influences tracheal tube morphogenesis was unexpected,” says Hayashi. “We speculate that the fat body plays a role in balancing the supply of a key enzyme to the trachea and epidermis so that differentiation of those epidermal organs is controlled under the same temporal schedule.”
| Link to article |
|---|